CLS Publishes Interim Report January – September 2025
14 november, 2025
14 november, 2025
CLS Publishes Interim Report January – September 2025
Lund, Sweden – Clinical Laserthermia Systems AB (publ) ("CLS" or the "Company") today publishes its interim report for January – September 2025.
Highlights of the Third Quarter Interim Report
During the third quarter, CLS continued to strengthen its operational and financial position while producing and delivering products, development, and support services for growing the adoption and installed base of its product portfolio.
Summary of the interim report (relates to the Group)
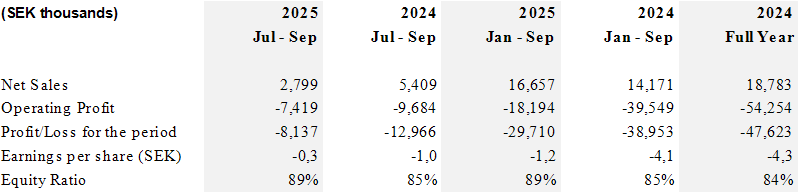
Comments from CEO Dan J. Mogren
In September, we achieved a key milestone when the U.S. Food and Drug Administration (FDA) granted expanded clearance for CLS ClearPoint Prism® branded Neuro Laser Therapy System to include both 3.0T and 1.5T MRI scanners. This approval more than doubles the addressable U.S. market for Neuro LITT procedures and positions CLS and ClearPoint Neuro for accelerated adoption in hospitals that previously could not access the technology.
The clearance represents not only an important commercial inflection point but also validation of the clinical versatility of our laser therapy platform. The first installations of the 1.5T labeled PRISM system are expected in the fourth quarter of this year.
In early October we announced positive results from the clinical safety study on laser ablation, performed using CLS Prism branded LITT platform in patients with malignant brain tumor. The study was conducted at Skåne University Hospital and presented in October at the CNS annual meeting in Los Angeles.
Patients in the study showed increased median survival compared to a matched control group treated with open surgery. We are very pleased that the primary clinical safety objectives of the study were successfully achieved and by positive feedback from the neurosurgeons that the system is easy to use with a reproducible workflow enabling a median ablation time of 6.5 minutes.
Further empowered by the recent successful regulatory and clinical study achievements, and in awareness of the current challenging trade climate, we have intensified conversations with our upstream and downstream partners to align on an accelerated growth scenario going forward.
In line with our growth strategy, the company has decided to actively start expanding the partnership model outside of neurosurgery. This decision aims to broaden the company’s commercial base and create a diversified, more balanced and predictable growth platform going forward.
Looking ahead, our priorities remain clear:
With strong clinical momentum, lowered barriers to adoption and market expansion, a focused organization, and a solid financial foundation, CLS is well positioned to deliver on its mission — to make minimally invasive laser therapy available to more patients worldwide.
Thank you for your continued support, and we look forward to your active participation in the exciting developments to come.
Dan J. Mogren
CEO, Clinical Laserthermia Systems AB
Significant events in the third quarter of 2025
Significant events after the end of the period
For more information, please contact:
Dan J. Mogren, CEO Clinical Laserthermia Systems AB (publ)
Phone: +46 (0)705 90 11 40
E-mail: dan.mogren@clinicallaser.com
About CLS
Clinical Laserthermia Systems AB (publ), develops and sells TRANBERG® Thermal Therapy System and ClearPoint Prism® Neuro Laser Therapy System with sterile disposables, for minimally invasive treatment of cancer tumors and drug-resistant epilepsy. The products are marketed and sold through partners for image-guided laser ablation. CLS is headquartered in Lund, Sweden, with subsidiaries in Germany, the United States and a marketing company in Singapore. CLS is listed on Nasdaq First North Growth Market under the symbol CLS B. Certified adviser (CA) is FNCA Sweden AB.
For more information about CLS, please visit the Company's website: www.clinicallaser.se
This disclosure contains information that CLS is obliged to make public pursuant to the EU Market Abuse Regulation (EU nr 596/2014). The information was submitted for publication, through the agency of the contact person, on 14-11-2025 08:30 CET.
14 november, 2025
CLS Publishes Interim Report January – September 2025
Lund, Sweden – Clinical Laserthermia Systems AB (publ) ("CLS" or the "Company") today publishes its interim report for January – September 2025.
Highlights of the Third Quarter Interim Report
During the third quarter, CLS continued to strengthen its operational and financial position while producing and delivering products, development, and support services for growing the adoption and installed base of its product portfolio.
Summary of the interim report (relates to the Group)
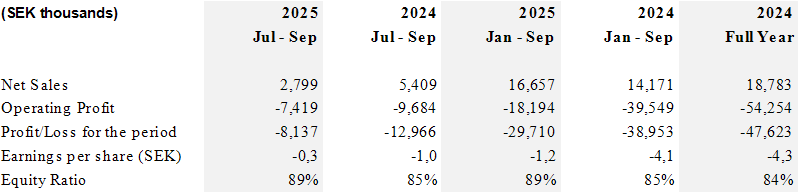
Comments from CEO Dan J. Mogren
In September, we achieved a key milestone when the U.S. Food and Drug Administration (FDA) granted expanded clearance for CLS ClearPoint Prism® branded Neuro Laser Therapy System to include both 3.0T and 1.5T MRI scanners. This approval more than doubles the addressable U.S. market for Neuro LITT procedures and positions CLS and ClearPoint Neuro for accelerated adoption in hospitals that previously could not access the technology.
The clearance represents not only an important commercial inflection point but also validation of the clinical versatility of our laser therapy platform. The first installations of the 1.5T labeled PRISM system are expected in the fourth quarter of this year.
In early October we announced positive results from the clinical safety study on laser ablation, performed using CLS Prism branded LITT platform in patients with malignant brain tumor. The study was conducted at Skåne University Hospital and presented in October at the CNS annual meeting in Los Angeles.
Patients in the study showed increased median survival compared to a matched control group treated with open surgery. We are very pleased that the primary clinical safety objectives of the study were successfully achieved and by positive feedback from the neurosurgeons that the system is easy to use with a reproducible workflow enabling a median ablation time of 6.5 minutes.
Further empowered by the recent successful regulatory and clinical study achievements, and in awareness of the current challenging trade climate, we have intensified conversations with our upstream and downstream partners to align on an accelerated growth scenario going forward.
In line with our growth strategy, the company has decided to actively start expanding the partnership model outside of neurosurgery. This decision aims to broaden the company’s commercial base and create a diversified, more balanced and predictable growth platform going forward.
Looking ahead, our priorities remain clear:
With strong clinical momentum, lowered barriers to adoption and market expansion, a focused organization, and a solid financial foundation, CLS is well positioned to deliver on its mission — to make minimally invasive laser therapy available to more patients worldwide.
Thank you for your continued support, and we look forward to your active participation in the exciting developments to come.
Dan J. Mogren
CEO, Clinical Laserthermia Systems AB
Significant events in the third quarter of 2025
Significant events after the end of the period
For more information, please contact:
Dan J. Mogren, CEO Clinical Laserthermia Systems AB (publ)
Phone: +46 (0)705 90 11 40
E-mail: dan.mogren@clinicallaser.com
About CLS
Clinical Laserthermia Systems AB (publ), develops and sells TRANBERG® Thermal Therapy System and ClearPoint Prism® Neuro Laser Therapy System with sterile disposables, for minimally invasive treatment of cancer tumors and drug-resistant epilepsy. The products are marketed and sold through partners for image-guided laser ablation. CLS is headquartered in Lund, Sweden, with subsidiaries in Germany, the United States and a marketing company in Singapore. CLS is listed on Nasdaq First North Growth Market under the symbol CLS B. Certified adviser (CA) is FNCA Sweden AB.
For more information about CLS, please visit the Company's website: www.clinicallaser.se
This disclosure contains information that CLS is obliged to make public pursuant to the EU Market Abuse Regulation (EU nr 596/2014). The information was submitted for publication, through the agency of the contact person, on 14-11-2025 08:30 CET.
Fonder
Analys
Bolåneräntorna
ETF:er
Fonder
Analys
Bolåneräntorna
ETF:er
1 DAG %
Senast
OMX Stockholm 30
2,63%
(vid stängning)
OMX Stockholm 30
1 DAG %
Senast
3 006,37